Clinical Outcome of AYAs with Head and Neck Sarcomas after Pencil Beam Scanning Proton Therapy
Miriam Vazquez,
Switzerland
MO-0379
Abstract
Clinical Outcome of AYAs with Head and Neck Sarcomas after Pencil Beam Scanning Proton Therapy
Authors: Miriam Vazquez1, Amaia Ilundain1, Dominic Leiser1, Barbara Bachtiary1, Alessia Pica1, Ulrike L Kliebsch1, Damien C Weber1
1Paul Scherrer Institut, Center for Proton Therapy, Villigen, Switzerland
Show Affiliations
Hide Affiliations
Purpose or Objective
Adolescents and young adults (AYAs) with cancer constitute a subgroup of patients that face unique challenges. Head and neck sarcomas (HNSs) are rare tumors that typically require aggressive multimodal treatment, including radiation therapy. Our aim is to assess the clinical outcome of AYAs with HNS treated with pencil beam scanning proton therapy (PBSPT).
Material and Methods
Twenty-eight AYAs (15-39 years) with HNSs treated between January 2001 and July 2021 at our institution were included in our analysis. Patients with skull-base tumors and mixed treatment with photons were excluded. The median age was 21.6 years (range, 15 - 37.9). Rhabdomyosarcoma (39.3%), Ewing sarcoma (17.9%), Chondrosarcoma (14.3%) and Osteosarcoma (14.3%) were the most frequent diagnosis. The median tumor at diagnosis was 42.5 mm (range, 12 - 95). Three (10.7%) patients were metastatic before the PBSPT. Five (17.9%) had a complete tumor resection, 11 (39.3%) a R1 resection, 4 (14.3%) a R0 resection and 8 (28.6%) had only a biopsy. The median total radiation dose was 63 Gy (RBE) (range, 45 - 74) delivered in a median dose per fraction of 1.8 Gy (RBE) (range, 1.8 - 2.2). Three (10.7%) patients were reirradiated. Thirteen (46.4%) received concomitant chemotherapy. Toxicity was reported according to Common Terminology Criteria for Adverse Events (CTCAE 5.0). Survival was estimated using the Kaplan–Meier method.
Results
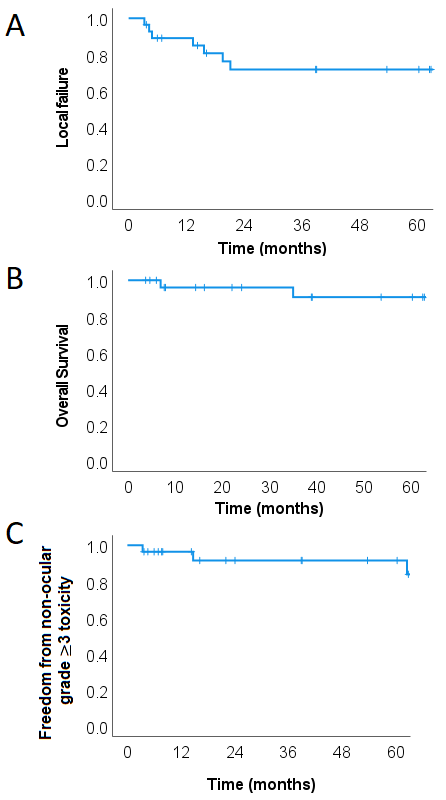
With a median follow-up of 57 months (range, 37 – 243), 4 patients (14.3%) had local failure only, 2 (7.1%) experienced distant failure only and 2 (7.1%) had local and distant failure. The estimated actuarial 5-year local control (LC) was 71.8% (Fig. 1A). Median time to local failure (LF) was 13.4 months. Three (10.7%) patients died, all of them from their cancer. Estimated 5-year overall survival (OS) was 90.9% (Fig. 1B). Six (21.4%) patients developed non-ocular grade ≥ 3 toxicity, which consisted of otitis media (n=2), hearing impairment (n=2), osteoradionecrosis (n=1) and sinusitis (n=1). Four (14.3%) patients developed cataract that required surgery. The 5-year freedom from non-ocular grade 3 toxicity was 91.1% (Fig. 1C). Median time to grade 3 toxicity was 66.1 months. No grade 4 or more toxicity was observed.
Conclusion
Excellent clinical outcomes with acceptable late toxicity rates were observed for AYAs with HN sarcomas after PBSPT.