Ultrahypofractionated Proton RT in Prostate Cancer: a single-center two-year experience
Nikita Kataev,
Russian Federation
PO-1505
Abstract
Ultrahypofractionated Proton RT in Prostate Cancer: a single-center two-year experience
Authors: Nikita Kataev1, Nikolay Vorobyov1, Alexei Mikhaylov1, Natalia Berezina2, Mikhail Cherkashin2, Vladislav Kovalik3
1Dr. Sergey Berezin Medical Institute (MIBS), Radiation Oncology, Saint Petersburg, Russian Federation; 2Dr. Sergey Berezin Medical Institute (MIBS), Administrative Department, Saint Petersburg, Russian Federation; 3ITMO University, Public Health, Saint Petersburg, Russian Federation
Show Affiliations
Hide Affiliations
Purpose or Objective
To analyze the biochemical disease-free survival (bDFS) and late toxicity profile in patients with prostate cancer treated with pencil beam scanning (PBS) proton radiation therapy in the first two years in our center.
Material and Methods
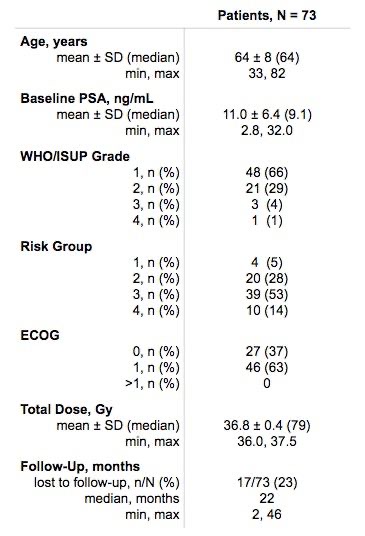
Between February 2019 and February 2021, 73 prostate cancer patients were treated using intensity-modulated proton therapy (IMPT) with an ultrahypofractionated schedule with a median total dose of 36.83 Gy SD=0.41 Gy in 5 fractions (range, 36-37.5 Gy). The mean age at the time of treatment was 63.9 SD=8.4 (range, 33-82) years, and the median prostate-specific antigen (PSA) value was 9.05 μg/L (2.83-32.00 μg/L).
A total of 25 (34.2%) patients had low-risk, 37 patients (50.7%) intermediate-risk, and 11 (15.1%) patients high-risk cancer. WHO/ISUP grade group was 1 in 48 (66.7%) patients, 2 in 21 (28.7%) patients , 3 in 3 (4.1%) and 4 in 1 (0.5%) patient. Of 73 patients, 17 were lost from follow-up and thus were excluded from the analysis. Data for 56 patients was collected by patient reports and analyzed with a median follow-up time of 22 (range, 2-46) months.
In addition, 9 (12.3%) patients underwent neoadjuvant hormonal therapy, and no patients had adjuvant hormonal therapy. Patient-reported PSA levels and prostate cancer radiotherapy late toxicity (PCRT) questionnaire results were obtained by phone.
Results
The median treatment time was 5.3 days (range, 5-8 days). There was no observed biochemical failure and clinical progression. Late toxicity (Common Terminology Criteria for Adverse Events v.5.0) was as follows: gastrointestinal: grade 1, 10 patients (17.9%), grade 2, 3 patients (5.3%), and no patients reported grade 3 or higher GI toxicity; genitourinary: grade 1, 8 patients (14.3%), grade 2, 6 patients (10.7%), and no patients reported grade 3 or higher GU toxicity. De novo ED disfunction rate was reported in 8 patients (14.3%).
Two patients (2.7%) died of causes unrelated to prostate cancer. No deaths related to prostate cancer were reported.
Conclusion
Ultrahypofractionated proton beam radiation therapy for prostate cancer is effective with long-term bDFS comparable with other fractionation schedules and treatment modalities Patients reported a low rate of GU, GI toxicity, and ED.