The steep dose gradient is one of the benefits of
brachytherapy as it enables delivery of high doses to the target while sparing
healthy tissues leading to excellent clinical outcome. At the same time, it
also puts high constraints on the treatment accuracy. This, together with the several
manual steps involved in brachytherapy, makes it important to perform treatment
delivery verification. The most direct way to verify the dose delivery is
through in vivo dosimetry, where the
dose is measured during delivery. The steep dose gradient puts high constraints
on both the internal and positional accuracy of any in vivo dosimetry system. Currently, there are no commercially
available system that has the sufficient accuracy and treatment delivery verification is not routinely available in
brachytherapy. This has led, in some cases, to systematic errors going
unnoticed for years.
In 2017, an ESTRO task group
was established to investigate how to enhance the clinical implementation of in vivo dosimetry in both external beam
radiotherapy and brachytherapy. This presentation will report on the findings
of the brachytherapy part of this task investigation.
it was found that the likelihood of detecting deviations from the treatment plan
increases significantly with time-resolved methods. Time–resolved methods could
interrupt a treatment avoiding gross errors which is not possible with
time-integrated dosimetry. In addition, lower experimental uncertainties can be
achieved by using more advanced techniques such as source-tracking instead of
direct dose measurements, fig. 1. However, the detector position in relation to
the patient anatomy remains a main source of uncertainty.
Several time-resolved systems have been developed and
tested in laboratories, but only few are used clinically. The development was
found to be driven by research groups and small start-up companies. These new
systems use different techniques such as point detectors, flat panels and
pin-hole detectors. All with their own pros and cons. It is of utmost
importance that the sensitivity to different types of errors is well understood
for each system as this will enable the end-users to select the most suitable
method for their needs.
The many new developments within dosimetry systems for
brachytherapy bodes well for the future use of in vivo dosimetry. The next steps towards clinical implementation
of in vivo dosimetry will require
clinical trials and systematic reporting of errors and near-misses.
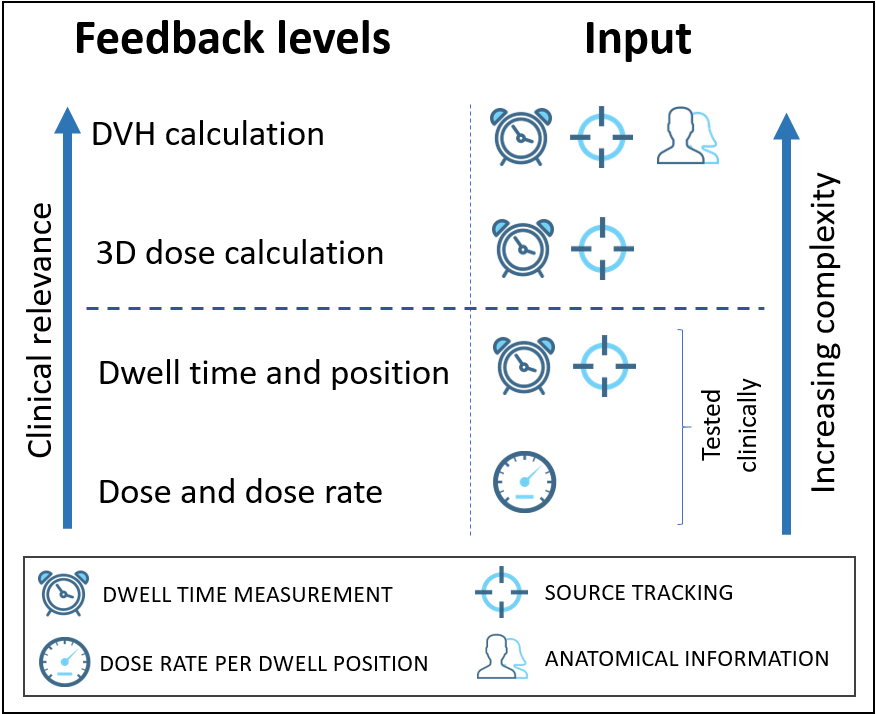
Figure
1: Feedback levels and corresponding necessary measurement inputs.