Objective:
To provide an overview on the multi-centre IntErnational studies on MRI-guided
BRAchytherapy in locally advanced CErvical Cancer (EMBRACE): the retrospective
RetroEMBRACE (RE), the prospective observational EMBRACE I (EI) and the
interventional/observational EMBRACE II (EII).
Materials/Methods:
RE, EI and EII provide comprehensive data from 731, 1416 and 284 pts (620
12/2019), respectively, with locally advanced cervical cancer (LACC) stage
IB-IVA and IVB (in paraaortic nodes (PAN)). Pts were treated before start of
EMBRACE in 12 centres (RE), from 2008-2015 in 23 centres (EI) and from 2016-12/2018
in 14 centres. Treatment was definitive EBRT (3DCRT or IMRT/IGRT) and
concurrent cisplatin followed by MR image guided adaptive brachytherapy (IGABT)
with MRI available for at least one fraction. IGABT targets, OAR and dose
volume parameters were reported following GEC-ESTRO recommendations (2005). In
RE and EI dose prescription followed institutional guidelines. EBRT dose was
45-50Gy (1.5-2.0Gy/fraction). In EII a multiparametric dose prescription
protocol is mandatory differentiating between soft and hard constraints for
targets (GTV, CTVHR, CTVIR)
and for OARs (D2cm3 for bladder,
rectum, sigmoid, bowel and rectovaginal point, bladder point (new vagina PIBS
concept)). A new tumor and risk adapted LN target concept was introduced
for IMRT in EII: CTV-T, CTV-E, ITV, PTV (45 Gy) and CTV-N, SIB (~60Gy); PA IMRT
was prescribed for high risk N+ pts. A comprehensive QA programme was applied
in EI and EII. Patient, tumor, treatment and outcome characteristics (crude
rates, actuarial estimates) are reported. Toxicity was prospectively assessed
in EI, EII (CTCAEv3.0).
Results:
Median age was 53, 56, 51 years and FIGO2009 stage
distribution was for RE, EI, EII: IB in 17,18,15%, IIB 50, 52, 57%, IIIB
20,14,18%, IVA 3,3,3%, IVB 6,7%; 40,52,58% were node-positive (N+); 85,82,81%
had squamous Ca, 9, 14, 16% adeno, 6, 4, 3% adenosquamous.
Mean EBRT dose was 46±2.5, 46±2, 45±1 Gy in RE, EI, EII. 3D CRT and IMRT was in
91 and 9% in RE, 59 and 41% in EI and IMRT/IGRT in 100% in EII. Para-aortic RT
was in 15, 17, 30% in RE, EI, EII. V43Gy was mean 2390/1418 cm3 in EI/EII for
pelvis and 2895/1765 cm3 in EI/EII for pelvis+PAN (Berger). 77, 95, 95%
received chemoth. IGABT technique was intracavitary alone (IC) in 77, 57, 27 %
and IC/interstitial (IS) in 23, 43, 73% in RE, EI, EII. Mean CTVHR volume
was 37±24, 33±19, 30±16 cm3 in RE, EI, EII: mean
CTVHR D90% was
overall 87±15, 89±9, 93±4 Gy10: for stage IB 93±17,
90±8, 93±3 Gy10; for stage IIB 88±14, 91±9, 93±4 Gy10;
for stage IIIB 83±13, 87±8, 93±4 Gy10;
for stage IVA 78±13, 83±10, 92±6 Gy10.
Isodose surface volumes were adapted to volume of CTVHR:
V85 Gy10 decreased
overall by 23% compared to Pt A Standards.
In RE, EI, EII, mean D2cm³ for bladder was
81±22, 76±10, 75±9 Gy3, rectum 64±9, 63±7,
59±6 Gy3,
sigmoid 66±10, 64±7, 62±7 Gy3, bowel 64±9, 63±10,
59±9 Gy3, rectovaginal
point 66±9, 62±7 Gy3.
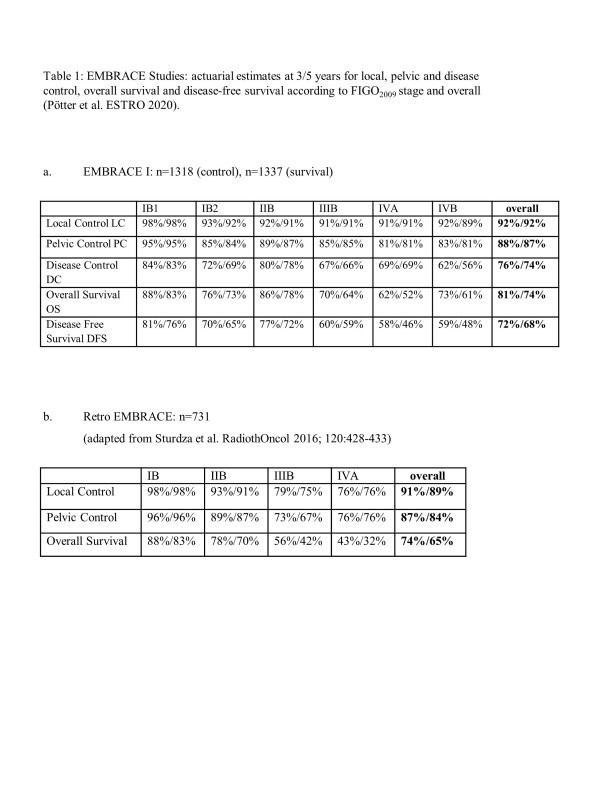
Median follow up was 43, 51 mths for 731, 1318 pats, resp. for RE, EI. Overall
30, 25% pts had recurrence (multiple events possible) in RE, EI: crude failure
rates were local 9.4, 7.4%; overall pelvic 13.1, 12.5%, nodal pelvic 5.6, 6.7%;
PAN 8.6, 8%. 21, 14% had recurrence beyond PAN. At the time of analysis, 35,
27% pts had died, 27, 21% from disease progression. In RE and EI at 5 years,
actuarial LC was 89, 92%, PC 84, 87%, DC 70, 74%, OS 65, 74%, CSS 73, 79%
(table 1). 5-year OS was 71,81% for N- pts and 57, 67% for N+ pts.
In RE and EI crude G3-5 morbidity at 5 years was genito-urinary 5, 6.5%,
gastro-intestinal 7, 7.6%, vagina 5, 6.1%.
Conclusion:
Age, local FIGO2009 stage and histology are comparable
within the EMBRACE studies. More nodal disease is diagnosed in EI and EII. EBRT
technique changed from predominant 3DCRT in RE to 100% IMRT/IGRT in EII with
markedly reduced volumes (V43 Gy) and increasing frequency of risk adapted PA
RT. Concomitant chemotherapy is increased in EI and EII. IC/IS techniques have
increased significantly during E1 and even more in EII which enables
appropriate target coverage and OAR sparing. CTVHR D90% is
overall increasing, in particular for large volumes. OAR D2cm3 are
being reduced in EII. Dose homogeneity is much increasing for targets and OARs
across patients.
From RE to EI overall local and pelvic control is excellent and even improving,
based on systematic use of advanced MRI based IGABT including interstitial
needles. Significant improvement is seen in stage IIIB and IVA. Para-aortic
recurrence remains similar. Overall survival and Cancer specific survival are
clearly improved.
Overall advanced MRI-based IGABT in combination with chemo-RT results in
excellent and stable long-term local, pelvic and overall disease control and
survival across all stages with limited severe morbidity. This EMBRACE
experience should be used as benchmark for routine clinical practice as well as
for design of any future clinical trials