Determinants of radioresistance and progression-free interval in SBRT-treated spinal metastases
PO-1444
Abstract
Determinants of radioresistance and progression-free interval in SBRT-treated spinal metastases
Authors: Michele Aquilano1, Sara Lucidi1, Loi Mauro2, Giulio Francolini3, Gabriele Simontacchi2, Daniela Greto2, Isacco Desideri4, Pierluigi Bonomo2, Andrea Gaetano Allegra1, Matteo Mariotti1, Laura Masi5, Raffaella Doro5, Ivano Bonucci6, Vanessa Di Cataldo6, Monica Mangoni1, Lorenzo Livi1
1University of Florence, Department of Experimental and Clinical Biomedical Sciences "M. Serio", Florence, Italy; 2Azienda Ospedaliero-Universitaria Careggi, Department of Radiation Oncology, Florence, Italy; 3Azienda Ospedaliero-Universitaria Careggi, Department of Radiation Oncology, Florence, Italy; 4University of Florence, Department of Experimental and Clinical Biomedical Sciences "M. Serio" , Florence, Italy; 5Radiation Oncology IFCA, Department of Medical Physics, Florence, Italy; 6Radiation Oncology IFCA, Department of Radiation Oncology, Florence, Italy
Show Affiliations
Hide Affiliations
Purpose or Objective
Stereotactic Body Radiotherapy (SBRT) is
increasingly used in uncomplicated spine metastases to palliate symptoms and
prolong disease control. However, criteria for patient selection are not
available. The aim of this study is to identify determinants of local failure
and progression-free interval in patients treated with SBRT to spinal
metastases.
Material and Methods
Data from consecutive patients treated with
Cyberknife-based spine SBRT between January 2019 and March 2020 were
retrospectively collected. Dose was expressed as Biological Effective Dose for α/β=10 (BED10). Kaplan-Meyer method was used to calculate Local
Control (LC) and Disease Progression-free Survival (DPFS) from date of SBRT to
event. Univariate (UVA) and Multivariate analysis (MVA) were performed using
log-rank and Cox model, respectively.
Results
Sixty-two patients accounting for 70 spinal
metastases were included. Median age was 66 (range 32-87) years. Disease was
metastatic at diagnosis in 21 patients (34%) : an active primary tumor was
present in 17 patients (27%). Among treated sites, most represented primary
malignancies were prostate (n=28, 40%) and breast (n=21, 30%). SBRT was
delivered to cervical, thoracic, lumbar and sacral vertebrae in respectively
15(21%), 21(30%), 29(41%), and 5(8%) cases. Dose regimens consisted of 25-30 Gy
in 5 fractions and 21-30 Gy in 3 fractions in respectively 61 (87%) and 9 (13%)
cases, resulting in a median BED of 43.2 (range 37.5-60) Gy10. Concurrent
chemotherapy (including cytotoxic or targeted agents) was administered in 43% of
cases (n=30). After a median follow up of 10 months (range 1-24 months), 9
local relapses and 40 distant progressions were observed. One year LC was 87% (Fig.1A): non-prostate
primary tumor ( p=0.003, Fig.1B) and concurrent chemotherapy (p=0.006, Fig.1C) were associated to
poorer LC at UVA, and an independent correlation was confirmed at MVA
(respectively p=0.017 and p=0.024). One-year DPFS was 43% (Fig.1D). UVA showed a
correlation between impaired DPFS and active primary tumor (p=0.003),
metastatic dissemination at diagnosis (p=0.02) and non-prostate primary tumor
(p=0.009), although only an active primary tumor site was independently
associated to DPFS at MVA (p=0.007, Fig.1E). Acute toxicity consisted of G2 pain flare and
G2 nausea in respectively 5 (7%) and 4 (6%) cases: no clinical variable was
significantly correlated with increased acute toxicity. No late toxicity, in
particular vertebral fracture, was reported.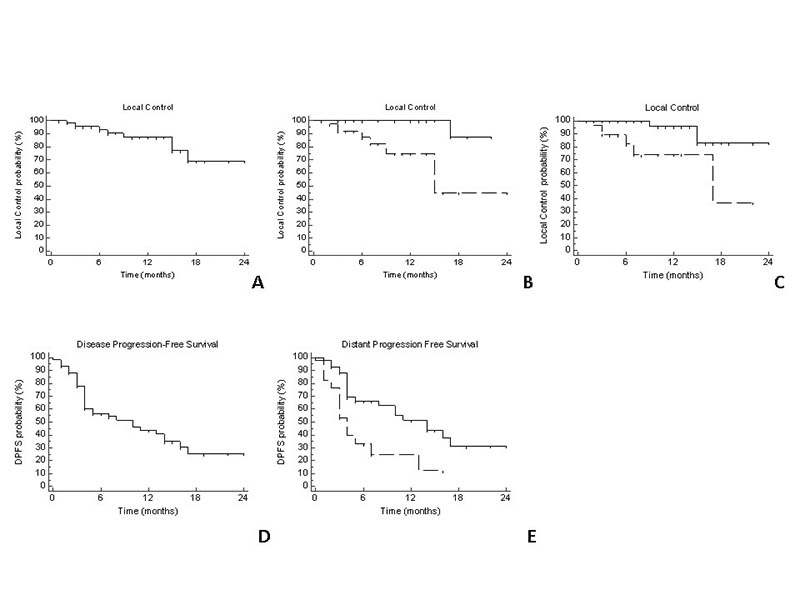
Conclusion
Spine SBRT results in high LC rates and durable
progression-free survival with modest incidence of mild toxicity. A better
knowledge of determinants of radioresistance (both intrinsically inherent to
tumor biology or acquired following repeated
administration of systemic therapy) is desirable due to potential correlation
with impaired LC. A controlled primary site is associated with longer time to
further distant progression following SBRT, supporting local treatment of
primary tumor in selected metastatic patients.