Modulated electro-hyperthermia plus systemic cancer treatment in digestive cancer. A safety study.
Elisabeth Estefanía Arrojo Alvarez,
Spain
PH-0555
Abstract
Modulated electro-hyperthermia plus systemic cancer treatment in digestive cancer. A safety study.
Authors: Elisabeth Estefanía Arrojo Alvarez1, Juan Carlos Mazabuel Quintero2, Diego Arribas Crespo2, Amaya Serrano Escalada2, Javier Anchuelo Latorre3
1Medical Institute of Advanced Oncology (INMOA) ; University Hospital Marqués de Valdecilla, Radiation oncology, Madrid; Santander, Spain; 2Medical Institute of Advanced Oncology (INMOA), Radiation Oncology, Madrid, Spain; 3University Hospital Marqués de Valdecilla, Radiation Oncology, Santander, Spain
Show Affiliations
Hide Affiliations
Purpose or Objective
Tumor vasculature is usually poor, narrow and aberrant, which can significantly limit oncological systemic treatment (OST) distribution and therefore effectiveness. This is usually more significant in more aggressive malignant tumors. Modulated electro-hyperthermia (mEHT), also known as oncothermia, has been proved as a selective type of hyperthermia for malignant tumors, which vasodilates only intratumoral vessels improving chemotherapy distribution inside the tumor, and minimizing the risk of other hyperthermias that create a general vasodilation in the treatment area, which could potentially increase the risk of distant metastases. Due to its selectivity, the risk of side effects from mEHT treatment is also very low. Up to date, there is only one phase III study with mEHT+chemotherapy and radiotherapy in cervical cancer patients with wonderful results. The purpose of this study, is to evaluate the safety of an mEHT treatment concomitant to OST (chemotherapy, immunotherapy, antiangiogenic therapy…) for digestive cancer.
Material and Methods
We retrospectively analyzed data from patients treated at the Medical Institute of Advanced Oncology (INMOA) between July 2019 and January 2021 for digestive carcinoma stage IV treated with OST plus mEHT applied at the abdominal area. CTCAE v5.0 grade scale was used for evaluating toxicity.
Results
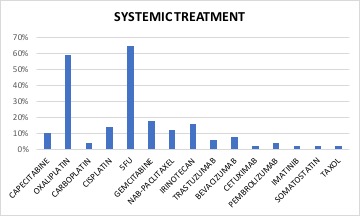
51 patients with stage IV cancer treated for digestive cancer with intraabdominal metastases were evaluated. Median age was 59 years (35-85). There were 25 females and 26 males. Most of the patients had colon (31%) or pancreatic cancer (31%), while 22% of the patients had gastric cancer, 6% cholangiocarcinoma, 4% esophagus, 4% rectal and 2% GIST. Metastatic site was liver in 80,4% of the patients and peritoneum in 20% of the patients. All patients were treated for at least 12 mEHT treatments and received at least 2 OST cycles concomitant to mEHT treatment. All mEHT treatments, were applied non consecutive days and had 90 minutes length. mEHT power range was between 80 and 150W.
None of the patients had skin burning. Grade 1-2 toxicities were developed in 96% of the patients, consisting of sickness, vomits, diarrhea and/or fever). Two patients had grade 3 toxicity (sickness and diarrhea) and needed to be admitted to the hospital, recovering some days later. One of these two, was under capecitabine treatment, and was diagnosed with a dihydropirimidin dehydrogenase deficit changing for this reason chemotherapy line. There were no grade 4 toxicities.
Conclusion
Modulated electro-hyperthermia concomitant to systemic oncological treatment was safe. mEHT is not a drug metabolized by the organism, and it is selective over malignant cells. These are probably some of the reasons why it does not increase toxicities from the systemic treatment. mEHT is a very promising treatment, as it can increase systemic and radiotherapy treatments effectiveness without increasing side effects for cancer patients.