Tumour reoxygenation by intratumoural hydrogen peroxide: a mechanism for enhanced radiosensitivity
Samantha Nimalasena,
United Kingdom
OC-0281
Abstract
Tumour reoxygenation by intratumoural hydrogen peroxide: a mechanism for enhanced radiosensitivity
Authors: Samantha Nimalasena1,2, Selva Anbalagan1, Carol Box1, Jessica K.R. Boult1, Nigel Bush1, Simon P. Robinson1, John Yarnold1, Navita Somaiah1,3
1The Institute of Cancer Research, Division of Radiotherapy and Imaging, London, United Kingdom; 2The Royal Marsden Hospital NHS Foundation Trust, Clinical Oncology, London, United Kingdom; 3The Royal Marsden NHS Foundation Trust, Clinical Oncology, London, United Kingdom
Show Affiliations
Hide Affiliations
Purpose or Objective
Following a Phase I trial confirming safety, a multicentre Phase II trial is underway comparing the efficacy of radiotherapy (RT) vs. intratumoural (IT) H2O2 + RT in breast cancer patients.
The hypothesis under test is that H2O2 leads to tumour reoxygenation, via breakdown into H2O and O2 within tumours. This is evidenced by O2 bubbles visible on ultrasound following IT injection. In vitro and in vivo studies were performed in a human colorectal tumour model to confirm the effect, and to investigate the reoxygenation hypothesis.
Material and Methods
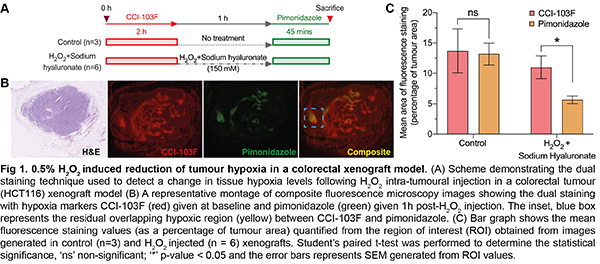
Subcutaneous HCT116 xenografts were established in Foxn1numice (n=12). Dual hypoxia markers were used to measure the difference in tumour hypoxia pre- and post- IT injection of 0.05ml 0.5% H2O2+ 1% sodium hyaluronate gel. IT injections were into the centre of the tumour under US guidance. A control cohort received no injection (n=3). CCI-103F was administered to provide a record of baseline tumour hypoxia, followed by pimonidazole (1 hour following IT injection). The experimental schema is shown in Fig.1A. A thresholding technique was used to quantify fluorescence on imaged tumour sections.
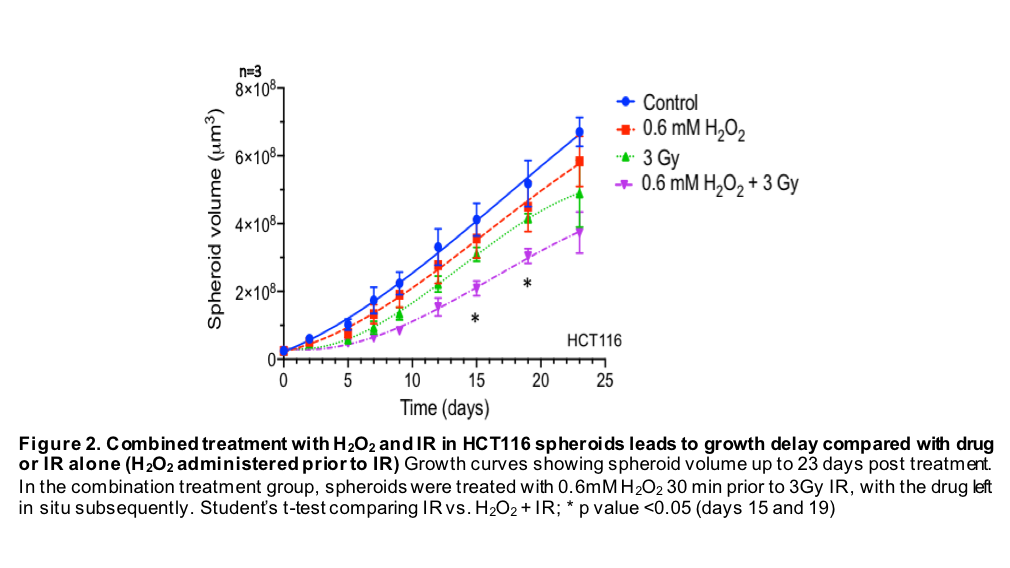
In order to test the combined effect of H2O2+ IR in a 3D in vitro model, HCT116 spheroids were treated with 0.6mM H2O2, 3Gy ionising radiation (IR), or a combination (0.6mM H2O2 administered 30 min pre-IR) and compared with untreated controls. Spheroid diameter was measured at 3-day intervals (until day 23) using an automated cytometer (Celigo) to monitor growth kinetics.
Results
HCT116 xenografts injected with H2O2 + sodium hyaluronate exhibited a reduction in hypoxia adduct formation (expressed as percentage of tumour area) from baseline (Fig.1B-C), in comparison to non-injected controls. A significant decrease (paired t-test) in hypoxia staining was demonstrated in H2O2-injected tumours (mean reduction 45%, range 25-61, p=0.02), compared to control tumours. Areas within the tumour showing abundant CCI-103F adducts were associated with less pimonidazole adducts following H2O2injection.
Growth inhibition was demonstrated in spheroids treated with the combination of H2O2+ IR, compared with H2O2 or IR alone, at all time points, and reaching statistical significance on day 15 and 19 (p=0.01) (Fig. 2).
Conclusion
Hypoxia quantification in HCT116 xenografts demonstrated significant reoxygenation following H2O2 injection. 3D spheroid data are in keeping with clinical reports demonstrating enhancement of radiation response by H2O2. Recapitulation of the clinical effect in vitro allows for further investigation of mechanisms of tumour response.
IT H2O2 represents a novel method of oxygen delivery and alleviating tumour hypoxia, which has implications for scheduling of H2O2 relative to RT in the clinic. If efficacy of IT H2O2+ RT is confirmed in the randomised phase II trial in breast cancer, it could improve treatment of several other tumour types where hypoxia is known to affect outcomes following RT.